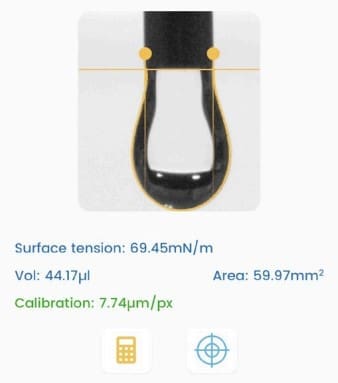
- The study used a Droplet Lab smartphone-based pendant drop tensiometer to quantify surface tension (aqueous–air) and interfacial tension (aqueous–mineral oil) via Young–Laplace / ADSA fitting (Methods, Section 2.5).
- These measurements were central for differentiating mechanisms:
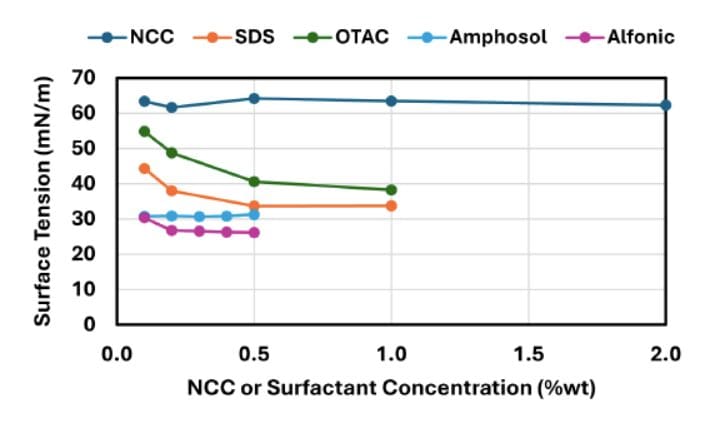
- NCC: minimal surface activity (little surface tension reduction), yet strong emulsion stabilization via interfacial jamming/steric stabilization.
- Surfactants (SDS, OTAC, Alfonic, Amphosol): significant reductions in surface/interfacial tension, aligning with concentration-dependent phase inversion behavior (Results, Section 3.2).
- Where the surface/interfacial tension measurements are mentioned: Methods Section 2.5 (pendant drop tensiometry details) and Results Section 3.2 (Surface Tension and Interfacial Tension; Figures 8–9).
- Contact angle note: The paper discusses “contact angle hysteresis” only as prior literature context; it does not report contact angle measurements in the experiments.